A case study
What is Vitamin D3?
Vitamin D is a fat-soluble prohormone that stimulates calcium absorption in the gut and maintains adequate serum calcium and phosphate concentration. Vitamin D2 and vitamin D3 are the main forms of Vitamin D used by humans. Vitamin D3 is produced naturally by the body when skin is exposed to ultraviolet radiation from the sunlight.
Deficiency of vitamin D may lead to diseases such as rickets in children, osteomalacia in adults, cancer, diabetes, and several chronic diseases.
EFSA has authorised Health Claims for Vitamin D regarding its role in the process of cell division, including its contribution to normal blood calcium levels, the maintenance of normal bones, the maintenance of normal muscle function, the maintenance of normal teeth, the normal function of the immune system, normal absorption/utilisation of calcium and phosphorous and its need for normal growth and development of bone in children.
FDA has authorised a Health Claim in the terms of Adequate calcium and vitamin D throughout life, as part of a well-balanced diet, may reduce the risk of osteoporosis.
Stability and Quality of Vitamin D3 Supplements
Vitamins easily degrade, influenced by a number of factors. In general, many physical and chemical factors can have a negative effect on the stability of vitamins. The most unstable vitamins are C, A, D, B1 and B12. Vitamin D3 is especially sensitive to light, oxidizing agents, heat, acidic and alkaline media.
As vitamin D3 stability is a non-solved issue, it is common practice to overdose vitamin D3 (technically called overage) to compensate the losses due to degradation. Obviously, the combination of degradation and overages can lead to high dispersion on the real vitamin D3 content with regard to the value claimed on the label of a dietary supplement.
For this reason, different Food Safety Agencies and associations have introduced limits to this practice. EFSA (European Food Safety Authority) allows a real vitamin D3 content between 80% to 150% of the valued claimed on the label; FDA (US Food and Drug Administration) requires a content between a 100% and a “reasonable” excess; RecHaN (Indian association) recommends overages of 30% for Vitamin D3.
Low Vitamin D3 stability leads to safety issues for end-users
Scientific studies carried out by independent researchers, in different countries, to compare the real Vitamin D3 content with the one claimed on the label, showed that the lack of stability of Vitamin D3 has a direct and relevant impact in the reliability of its label claim in supplements:
| TERRITORY | COUNTRY | Min Vit D3 content (vs label) | Max Vit D3 content (vs label) | Reference |
|---|---|---|---|---|
| US | 9% | 140% | (2013, Leblanc ES) | |
| 7% | 172% | (2013, Andrews KW) | ||
| Europe | Poland | 107% | 138% | (2000, Klackow G) |
| The Netherlands | 8% | 177% | (2017, Verkaik-Kloosterman J | |
| Slovenia | 100% | 153% | (2016 Temova Z) | |
| Slovenia | 65% | 156% | (2018 Temova Z) | |
| India | 9% | 165% | (2013, Khadgawat R) | |
| New Zealand | 8% | 201% | (2013, Garg S) | |
| Jordania | 5% | 94% | (2010, Al-Quadi E) |
Taking into account the relevance of vitamin D3 in the human health, it is hard to accept that a consumer buying a supplement claiming 20 micrograms of Vitamin D3 (the Recommended Daily Intake), can actually be taking between 1.6 and 40 micrograms per capsule (instead of the claimed 20 micrograms). Both a deficiency and an excess of vitamin D3 can have a negative impact on the consumer’s health. Thus, the lack of stability leads to a lack of quality in Vitamin D3-containing supplements. This unresolved technical limitation ultimately affects the reliability of the product.
“The determined vitamin D content was higher than stated on the label in most of the tested preparations, reaching up to 156% in our study and up to 200% in the published data. Even though the use of vitamin D overages is a routine, it poses a serious danger of excessive vitamin D intakes. These can be responsible for toxic effects, mediated mostly through hypercalcaemia.”
(Temova Rakuša, Ž et al. Vitamin D in Supplements and Medicines. Chapter 4. 2018)
In addition to the quality and safety issues related to this overage practice, it is clearly a non-sustainable policy to add a huge excess of Vitamin D3 in each capsule/pill, resignedly assuming it will inevitably degrade.
How do Cocrystals of Vitamin D3 overcome the safety issue associated with stability of Vitamin D3
CIRCE Scientific has discovered new cocrystals of Vitamin D3 that significantly improve its stability. On the one hand, the cocrystal showed much higher stability to light (80% retention after 2 months under 5,000 Lux lighting, vs. 20% retention of commercial Vitamin D3).
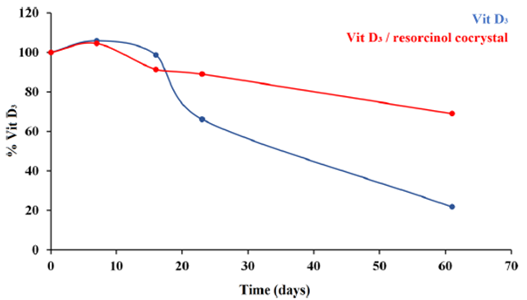
Under stress conditions (40ºC, 75% Relative Humidity, regular Vitamin D3 showed degradation after 2 weeks, while vitamin D3 remained stable after 2 months.
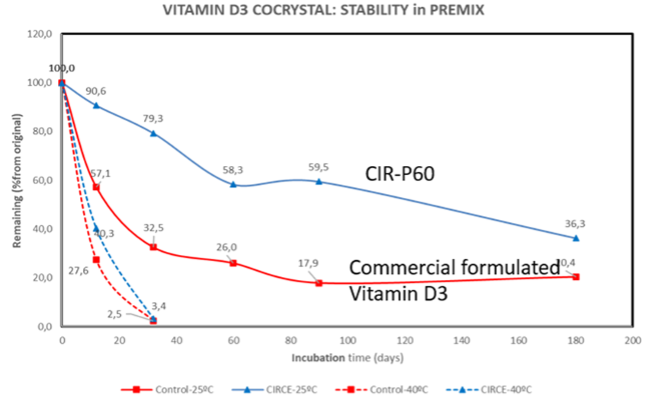
Finally, an experiment performed in collaboration with the Autonomous University of Barcelona showed that in a premix containing Vitamin D3, other vitamins, minerals and choline chloride (the worst-case scenario regarding degradation of Vitamin D3), CIRCE Scientific cocrystal was significantly more stable than a commercial Vitamin D3 formulated to increase its stability. After 3 months, the cocrystal retained 60%, while the commercial formulated Vitamin D3 retained just 18%.





