Stilbenes are phytochemicals, a subclass of polyphenolic compounds. They are naturally found in a wide variety of plant based dietary sources such as grapes, blueberries, red wine, and some other plants.
Among the well-known stilbenes are resveratrol, which is the first discovered and likely the most famous, and pterostilbene, which was discovered later and shows promise as an improvement over resveratrol.
Resveratrol vs Pterostilbene: Which one is the better stilbene?
Extensive research has been conducted on resveratrol and has demonstrated its potential health benefits, including the prevention of several adverse processes such as oxidative stress, inflammation, cancer, and diabetes.
However, Resveratrol’s potential biological effectiveness may be limited due to its very low bioavailability. For this reason, there are many products on the market with high dosis of this compound, reaching up to 1.5 g of resveratrol per dose.
For a stilbene to be effective, it must be absorbed in sufficient quantity and remain unaltered long enough to reach its target effect. However, resveratrol is rapidly metabolized, leading to minimal levels of unaltered resveratrol in blood after its intake.
Pterostilbene is an emerging stilbene showing similar properties to resveratrol, but without the handicap of a low bioavailability, making it a promising alternative to resveratrol.
It is more resistant to metabolic modifications, allowing a higher fraction of ingested pterostilbene to reach its targets and be available for biological activity compared to resveratrol.
While resveratrol has numerous potential health benefits, its low bioavailability has been a limitation that requires high doses to achieve the desired effects. Pterostilbene, with its superior bioavailability and activity (ORAC value Resveratrol 28 µmol Trolox equivalents per gram (TE/g), ORAC value Pterostilbene 64 µmol Trolox equivalents per gram (TE/g)) may be a better alternative for more efficient formultations.
Similar structures with one critical difference
Resveratrol and pterostilbene share a very similar chemical structure, both composed of two aromatic rings with hydroxyl groups (-OH). However, the crucial difference between them is that resveratrol has three hydroxyl groups, whereas pterostilbene only has one.
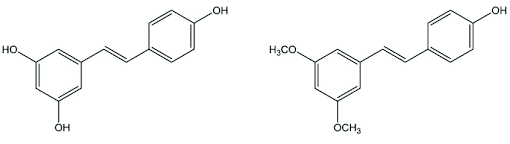
The three hydroxyl groups in resveratrol hasten the molecule’s removal, which makes it challenging to reach and maintain appreciable levels of resveratrol in the bloodstream.
In pterostilbene the other two hydroxyl groups are replaced by methoxy groups (O-CH3). This difference is crucial because it influences how quickly the compound is metabolized and eliminated from the body.
Due to its structural characteristic, pterostilbene is more lipophilic, exhibits better bioavailability, and is more biologically active than resveratrol.
Pterostilbene shows better results in studies
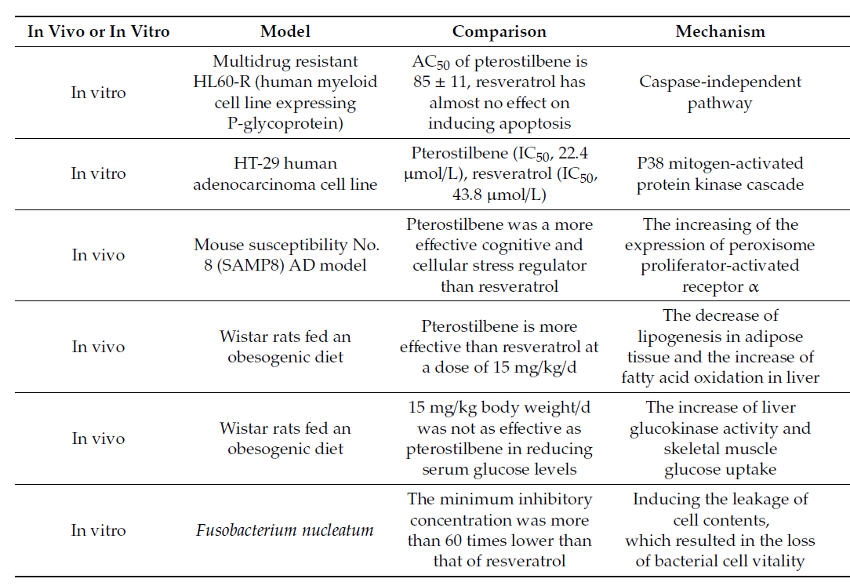
Some investigation in animals have shown that pterostilbene exhibits a superior pharmacokinetic profile compared to resveratrol, which could explain why pterostilbene’s pharmacological activity is often found to be superior to that of resveratrol.
Several publications have currently compared pterostilbene and resveratrol in various diseases and health conditions, including weight control and cognitive protection. In each of these publications, pterostilbene has demonstrated better results at similar concentrations compared to resveratrol.
All the studies show that Pterostilbene is significantly more bioavailable than resveratrol, however due to its low solubility, the body still absorbs a low fraction of the ingested product.
To overcome this issue, CIRCE Scientific has developed a highly improved version of Pterostilbene, PTEROVITA. Contact us to discover more.